SOLVED:Calculate the pH and the pOH of each of the following solutions at 25^∘ C for which the substances ionize completely: (a) 0.000259 MHClO4 (b) 0.21 MNaOH (c) 0.000071 MBa(OH)2 (d) 2.5 MKOH
![How to calculate the pH, pOH, [H+] & [OH-] of strong acids - YouTube | Ap chemistry, Chemistry, Biochemistry How to calculate the pH, pOH, [H+] & [OH-] of strong acids - YouTube | Ap chemistry, Chemistry, Biochemistry](https://i.pinimg.com/564x/7e/07/f6/7e07f6adc8c7363b328f561729f174ba.jpg)
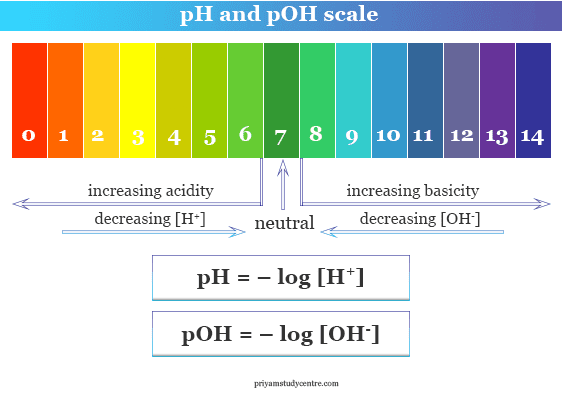
How to calculate the pH, pOH, [H+] & [OH-] of strong acids - YouTube | Ap chemistry, Chemistry, Biochemistry
![SOLVED:Calculate the pH and pOH of aqueous solutions with the following concentrations at 298 K . a. [OH^-]=1.0 ×10^-6 M b. [OH^-]=6.5 ×10^-4 M c. [H^+]=3.6 ×10^-9 M d. [H^+]=2.5 ×10^-2 M SOLVED:Calculate the pH and pOH of aqueous solutions with the following concentrations at 298 K . a. [OH^-]=1.0 ×10^-6 M b. [OH^-]=6.5 ×10^-4 M c. [H^+]=3.6 ×10^-9 M d. [H^+]=2.5 ×10^-2 M](https://cdn.numerade.com/previews/ae8f1499-088d-4c58-b4bd-3f0a48e0135e_large.jpg)
SOLVED:Calculate the pH and pOH of aqueous solutions with the following concentrations at 298 K . a. [OH^-]=1.0 ×10^-6 M b. [OH^-]=6.5 ×10^-4 M c. [H^+]=3.6 ×10^-9 M d. [H^+]=2.5 ×10^-2 M

![Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC" Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC"](http://itsactuallyquitebasic.weebly.com/uploads/2/7/8/0/27808159/4950515.png?357)
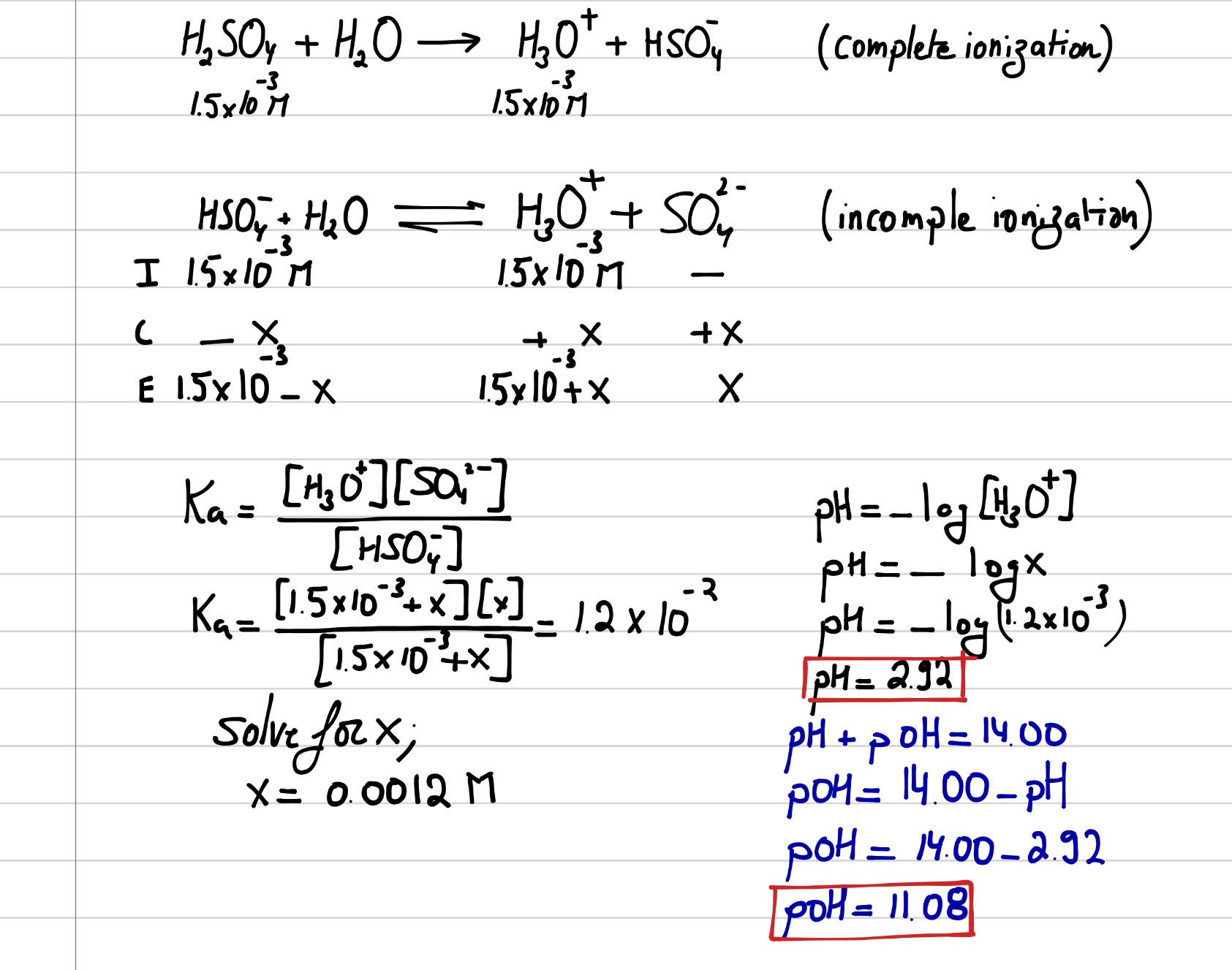



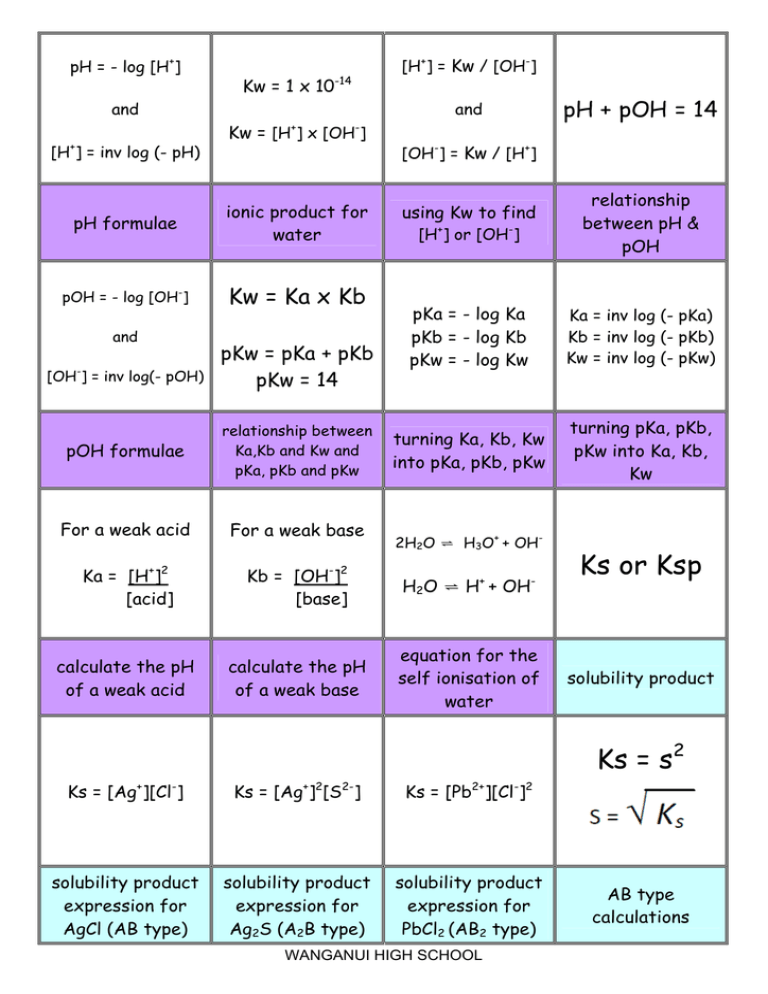






![Calculating [OH-], pH and pOH from Kb Calculating [OH-], pH and pOH from Kb](https://www.mi.mun.ca/users/pfisher/chemistry1011_135/img007.gif)
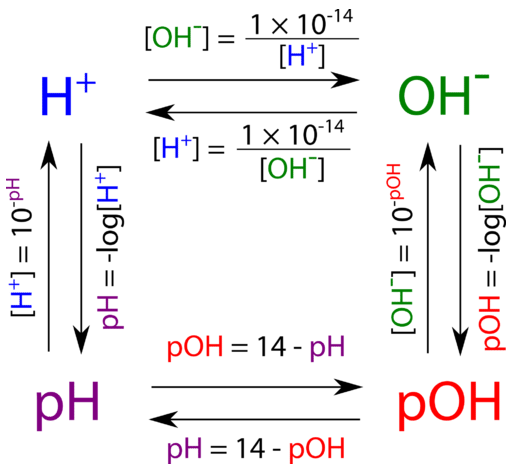
![Calculations of pH, pOH, [H+] and [OH-] Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)
![Calculating pH & pOH, [H+], [OH-], Acids & Bases CLEAR & SIMPLE - YouTube Calculating pH & pOH, [H+], [OH-], Acids & Bases CLEAR & SIMPLE - YouTube](https://i.ytimg.com/vi/eoM1nzYFy8k/maxresdefault.jpg)