Calculate the enthalpy change for the process CCl4(g)→ C(g) + 4Cl(g) and calculate bond enthalpy of C - Cl in CCl4(g) Δ vapH^ (CCl4) = 30.5 kJ mol ^-1 . Δ fH^ (

Calculate the enthalpy change for the reaction CH2 = CH2(g) + H2(g0 to CH3 - CH3(g). The bond energies of C - C , C - H, H- H and C =

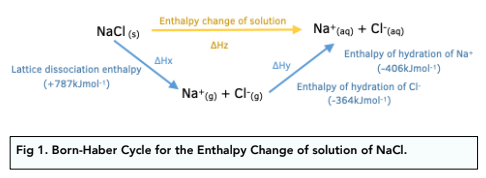
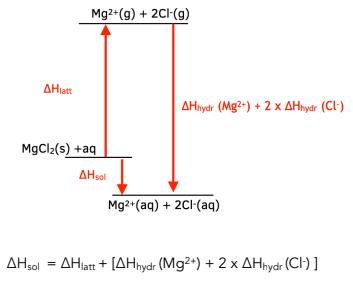
thermodynamics - Does the enthalpy of solution formula (LE + Hyd) change depending on the question? - Chemistry Stack Exchange

Calculate the enthalpy change for the process CCl4(g)→ C(g) + 4Cl(g) and calculate bond enthalpy of C - Cl in CCl4(g) Δ vapH^ (CCl4) = 30.5 kJ mol ^-1 . Δ fH^ (

Physical Chemistry #16: Calculating Enthalpy Changes and Calorimetry (Slides and Student Led Tasks) | Teaching Resources