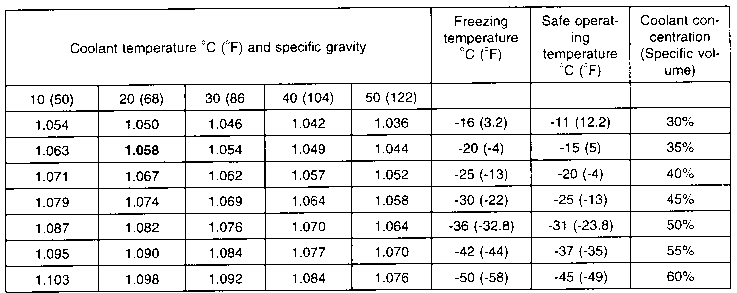
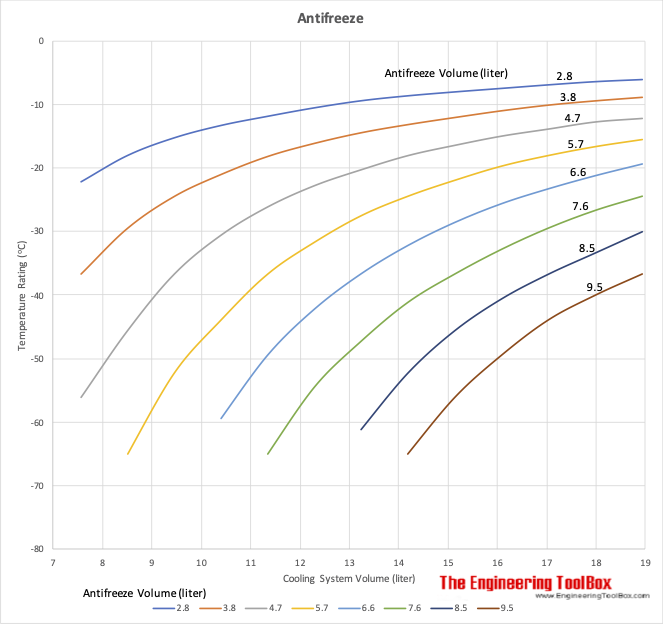
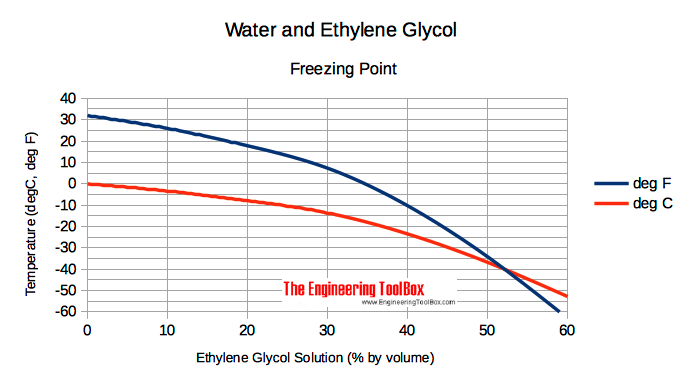
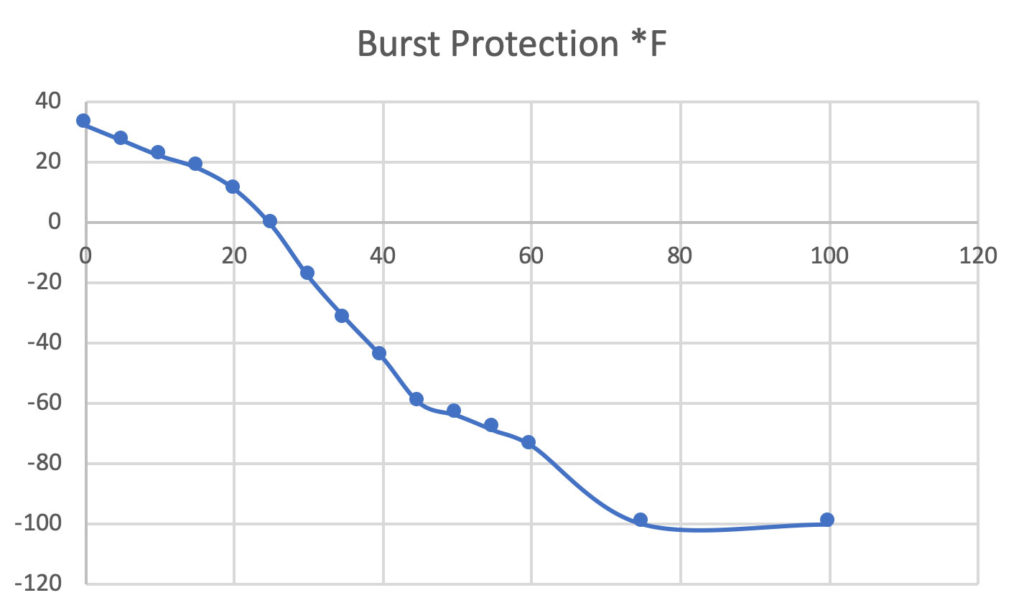
Ethylene glycol is used as an antifreeze agent. Calculate the amount of ethylene glycol to be added to 4 kg of water to prevent it from freezing at - 6^∘ C.(Kf for

SOLVED:Calculate the freezing point and boiling point of an antifreeze solution that is 50.0 % by mass of ethylene glycol (HOCH2 CH2 OH) in water. Ethylene glycol is a nonelectrolyte.

Antifreeze Refractometer Displaying in Fahrenheit for Checking Freezing Point of Automobile Antifreeze Systems and Battery Fluid Condition. Battery Acid, Glycol, Coolant, Antifreeze Tester: Amazon.com: Industrial & Scientific
![SOLVED: How many liters of the antifreeze ethylene glycol [CH2(OH)CH2(OH)] would you add to a car radiator containing 6.50 L of water if the coldest winter temperature in your area is 220°C? SOLVED: How many liters of the antifreeze ethylene glycol [CH2(OH)CH2(OH)] would you add to a car radiator containing 6.50 L of water if the coldest winter temperature in your area is 220°C?](https://cdn.numerade.com/ask_previews/9c9724b5-5961-4b32-a05e-7ba03745d4c6_large.jpg)
SOLVED: How many liters of the antifreeze ethylene glycol [CH2(OH)CH2(OH)] would you add to a car radiator containing 6.50 L of water if the coldest winter temperature in your area is 220°C?